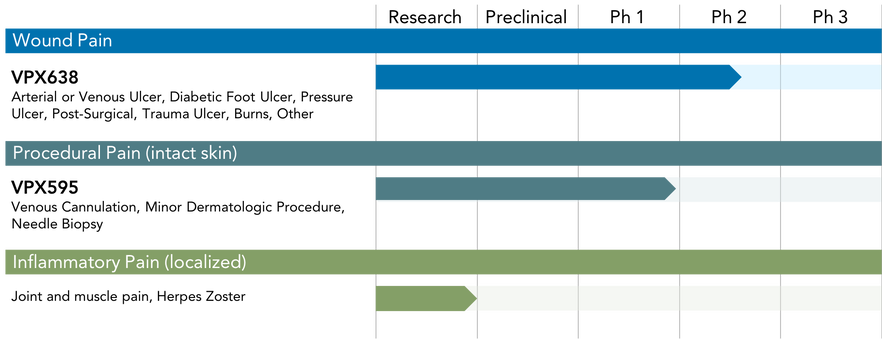
Pipeline
Vapogenix is developing a suite of products to treat localized pain using formulated volatile anesthetics.
Vapogenix is developing a suite of products to treat localized pain using formulated volatile anesthetics.
Wound Pain
Pain from wounds is widespread, yet frequently underestimated and poorly treated. The prevalence of non-healing wounds among Medicare beneficiaries alone is estimated at 8 million patients, indicating an even higher overall national burden. Current therapies used to address wound pain, including opioids, are almost exclusively systemic, often provide inadequate relief and can result in intolerable adverse effects as well as abuse and addiction.
Topical, non-opioid options for the management of painful wounds represent an area of significant unmet need in pain management
Vapogenix’s lead product, VPX638, is a novel, non-opioid topical analgesic in Phase 2 for the treatment of painful wounds. As a topical analgesic expected to offer rapid and long-lasting pain relief with an attractive safety profile, VPX638 has the potential to transform the management of wound pain
Vapogenix has successfully completed a randomized, well-controlled Phase 2 study of lead product VPX638 for the treatment of wound pain. Encouraging Phase II results will inform the design of the Phase III study.
Procedural Pain (intact skin)
The goal of this program is to develop a rapidly-acting topical analgesic for use on intact skin to alleviate pain acute procedural pain such as that associated with venipuncture, venous cannulation and minor dermatological procedures. Most of the currently available options require long application times, ranging from 20 minutes to 60 minutes. There is a huge unmet need for a rapidly-acting and effective topical analgesic, particularly for children and needle-phobic adults. We have completed a Phase I clinical study that demonstrated a promising safety and tolerability profile of our products.
Inflammatory Pain (localized)
Inflammation is associated with many acute and chronic painful conditions, such as arthritis, gout and muscle pain. Much of the pain associated with these conditions is due to the release of pain-inducing mediators from white blood cells at the site of inflammation. Some volatile anesthetics have anti-inflammatory effects, in addition to their analgesic properties, and therefore have the potential to be highly effective in painful inflammatory conditions. The first product under this program is designed to have a dual benefit, combining these analgesic and anti-inflammatory effects in a topical product.
Pain from wounds is widespread, yet frequently underestimated and poorly treated. The prevalence of non-healing wounds among Medicare beneficiaries alone is estimated at 8 million patients, indicating an even higher overall national burden. Current therapies used to address wound pain, including opioids, are almost exclusively systemic, often provide inadequate relief and can result in intolerable adverse effects as well as abuse and addiction.
Topical, non-opioid options for the management of painful wounds represent an area of significant unmet need in pain management
Vapogenix’s lead product, VPX638, is a novel, non-opioid topical analgesic in Phase 2 for the treatment of painful wounds. As a topical analgesic expected to offer rapid and long-lasting pain relief with an attractive safety profile, VPX638 has the potential to transform the management of wound pain
Vapogenix has successfully completed a randomized, well-controlled Phase 2 study of lead product VPX638 for the treatment of wound pain. Encouraging Phase II results will inform the design of the Phase III study.
Procedural Pain (intact skin)
The goal of this program is to develop a rapidly-acting topical analgesic for use on intact skin to alleviate pain acute procedural pain such as that associated with venipuncture, venous cannulation and minor dermatological procedures. Most of the currently available options require long application times, ranging from 20 minutes to 60 minutes. There is a huge unmet need for a rapidly-acting and effective topical analgesic, particularly for children and needle-phobic adults. We have completed a Phase I clinical study that demonstrated a promising safety and tolerability profile of our products.
Inflammatory Pain (localized)
Inflammation is associated with many acute and chronic painful conditions, such as arthritis, gout and muscle pain. Much of the pain associated with these conditions is due to the release of pain-inducing mediators from white blood cells at the site of inflammation. Some volatile anesthetics have anti-inflammatory effects, in addition to their analgesic properties, and therefore have the potential to be highly effective in painful inflammatory conditions. The first product under this program is designed to have a dual benefit, combining these analgesic and anti-inflammatory effects in a topical product.